Lipids Are a Family of Compounds That Include
5.ane: Introduction to Lipids
- Page ID
- 21122
Learning Objectives
- Depict the three types of lipids.
- Compare and contrast the structure of the following types of fats:
- triacylglycerols (including saturated, monounsaturated, polyunsaturated, cis, trans, omega-iii, and omega-vi fatty acids)
- phospholipids
- sterols
Does it surprise you to know that you lot should not eat a fat-free diet? Lipids are essential nutrients and we need these compounds in the nutrition in moderate amounts. Lipids are a family unit of organic compounds that are mostly insoluble in water. Composed of fats and oils, lipids are molecules that yield high energy and have a chemical limerick mainly of carbon, hydrogen, and oxygen. The three main types of lipids are triacylglycerols, phospholipids, and sterols (Effigy \(\PageIndex{one}\)).
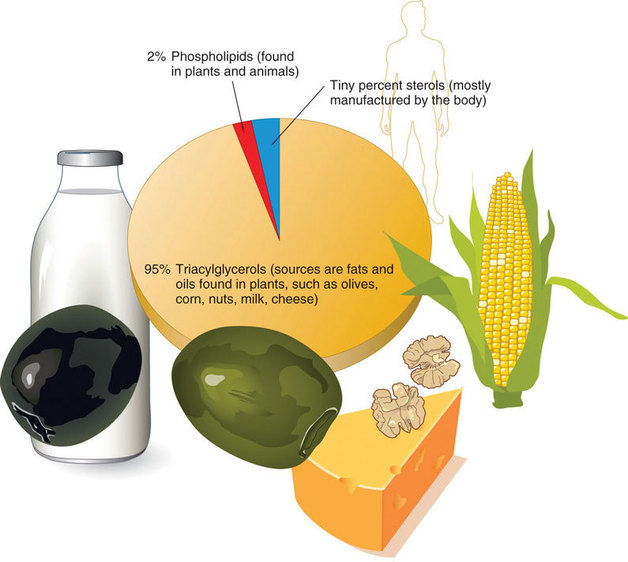
Triacylglycerols
Triacylglycerols (besides known as triglycerides) make upwards more than 95 pct of lipids in the nutrition and are normally found in fried foods, vegetable oil, butter, whole milk, cheese, cream cheese, and some meats. Naturally occurring triacylglycerols are institute in many foods, including avocados, olives, corn, and nuts. We usually call the triacylglycerols in our food "fats" and "oils." Lipids take two main forms - fats, which are solid at room temperature, and oils, which are generally liquid at room temperature. As with most fats, triacylglycerols do non deliquesce in water. The terms fats, oils, and triacylglycerols are often used interchangeably. In this chapter when we apply the word fat, we are referring to triacylglycerols.
Triacylglycerols (triglycerides) are the nigh common type of fat eaten and stored in the body. The types of fatty acids in our diet direct correlate to the types of fatty acids that are stored in our body.
Fatty acids and glycerol are the edifice blocks of triacylglycerols. To form a triacylglycerol, a glycerol molecule is joined by three fat acid bondage (Figure \(\PageIndex{2}\)). Glycerol is a 3-carbon alcohol that is the backbone of a triacylglycerol; diverse fat acids attach to the glycerol backbone.

Fatty Acids
Fat acids determine if the compound is solid or liquid at room temperature. Fat acids consist of a carboxyl group (−COOH) group on one end of a carbon chain and a methyl group (−CHthree) on the other cease (Effigy \(\PageIndex{iii}\)). Fatty acids can differ from one some other in 3 of import means—carbon chain length, degree of saturation, and shape.
Carbon Concatenation Length
Fat acids take different concatenation lengths and unlike compositions. The length of the carbon chain determines the method of digestion, assimilation, and send in the body. Foods accept fat acids with chain lengths betwixt four and twenty-four carbons and most of them incorporate an even number of carbon atoms. Fat acids can exist named based on their concatenation length; for instance, short- (less than vi carbons), medium- (6-12 carbons), or long-chain (14 or more carbons). When the carbon chain length is shorter, the melting point of the fatty acid becomes lower—and the fatty acrid becomes more than liquid.
Degree of Saturation
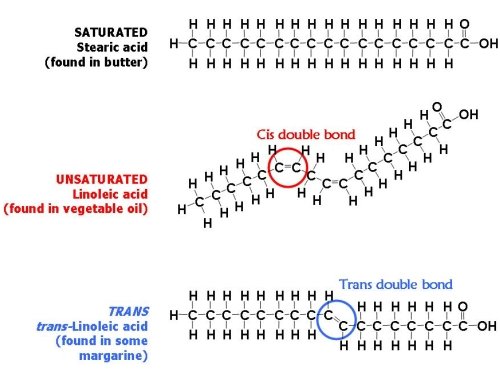
Fat acid bondage are held together by carbon atoms that attach to each other and to hydrogen atoms. The term saturation refers to whether or not a fat acid chain is filled (or "saturated") to capacity with hydrogen atoms. If each available carbon bond holds a hydrogen atom information technology'south a saturated fatty acrid chain (Figure \(\PageIndex{four}\)). All of the carbon atoms in a saturated fatty acrid chain are bonded through single bonds. The structure created by unmarried bonds means saturated fatty acids can pack tightly together and are solid at room temperature. Examples of foods high in saturated fatty acids include animal fats, butter, and lard.
If there is a place on the concatenation where a hydrogen atom is missing, it is known as the point of unsaturation. Unsaturated fatty acids do not stack together well and are liquid at room temperature. When one or more bonds betwixt carbon atoms are a double bond (C=C), that fatty acid is called an unsaturated fatty acid, every bit it has 1 or more points of unsaturation (Effigy \(\PageIndex{four}\)). Whatsoever fatty acrid that has only one double bail is a monounsaturated fatty acrid, an example of which is olive oil (75 percent of its fat is monounsaturated). Monounsaturated fats assistance regulate blood cholesterol levels, thereby reducing the risk for heart affliction and stroke. A polyunsaturated fatty acid is a fatty acid with 2 or more double bonds or two or more than points of unsaturation. Soybean oil contains loftier amounts of polyunsaturated fatty acids. Both monounsaturated fats and polyunsaturated fats provide diet that is essential for normal prison cell development.

Foods that have a high percentage of saturated fatty acids tend to be solid at room temperature. Examples of foods high in saturated fats include chocolate and meat. Foods rich in unsaturated fatty acids, such every bit olive oil, tend to exist liquid at room temperature.
Shape
The introduction of a carbon double bond in a carbon chain, as in an unsaturated fatty acid, can result in dissimilar structures for the same fat acid composition. When the hydrogen atoms are bonded to the same side of the carbon chain, it is called a cis fatty acid (Effigy \(\PageIndex{5}\)). Because the hydrogen atoms are on the same side, the carbon chain has a bent structure. Naturally occurring fat acids ordinarily have a cis configuration.
In a trans fatty acid, the hydrogen atoms are attached on opposite sides of the carbon chain (Figure \(\PageIndex{v}\)). Unlike cis fatty acids, virtually trans fatty acids are non found naturally in foods, only are a result of a process called hydrogenation. Hydrogenation is the process of adding hydrogen atoms to an unsaturated fat (the hydrogen atoms are added where there are double bonds), thus making the fat acid saturated (or less unsaturated, in the case of partial hydrogenation). This is how vegetable oils are converted into semisolid fats for apply in the manufacturing process. The hydrogenation process makes the fat more similar to a saturated fat and makes the fatty more stable which is advantageous for the shelf-life of products that include trans fats. Notwithstanding, trans fatty acids take been associated with increased risk for eye disease because of the way they negatively affect blood cholesterol levels. Dietary Guidelines recommend keepingtrans fat intake as low as possible. The Diet Facts console includes trans fat as a separate line item under the "Total Fat" section. In addition, ingredients that include the give-and-take "hydrogenated" or "partially hydrogenated" indicate the presence of trans fat in the food.
Knowing the connection between chain length, degree of saturation, and the shape of the fatty acid is important for making nutrient choices. If you decide to limit or redirect your intake of fat products, and so choosing unsaturated fat is more beneficial than choosing a saturated fatty. This choice is easy enough to make considering unsaturated fats tend to be liquid at room temperature (for example, olive oil) whereas saturated fats tend to exist solid at room temperature (for example, butter). Avocados are rich in unsaturated fats. Most vegetable and fish oils contain high quantities of polyunsaturated fats. Olive oil and canola oil are besides rich in monounsaturated fats. However, non all oils are unsaturated - tropical oils like palm oil and coconut oil are liquid at room temperature all the same high in saturated fat. Trans fat is associated with an increased risk for center disease; fortunately, trans fats have been eliminated from well-nigh processed foods.
Nonessential and Essential Fatty Acids
Fat acids are vital for the normal functioning of all body systems. The body is capable of synthesizing well-nigh of the fat acids information technology needs from food. These fatty acids are known every bit nonessential fat acids. However, in that location are some fatty acids that the body cannot synthesize and these are called essential fatty acids. Simply considering a fatty acid is classified as nonessential, information technology doesn't mean that it's non important; information technology but ways that the body is able to synthesize the fatty acid.
Essential fatty acids must be obtained from food. They fall into two categories—omega-3 and omega-six. The 3 and 6 refer to the position of the first carbon double bond from the omega (methyl) end of the chain. Omega-3 and omega-vi fatty acids are precursors to of import compounds called eicosanoids. Eicosanoids are powerful hormones that control many other hormones and important body functions, such as the central nervous system and the immune system. Eicosanoids derived from omega-6 fatty acids are known to increase blood pressure, immune response, and inflammation. In contrast, eicosanoids derived from omega-3 fatty acids are known to have middle-healthy effects. Given the contrasting effects of the omega-3 and omega-6 fatty acids, a proper dietary balance between the ii must exist achieved to ensure optimal health benefits.
Alpha-linolenic acid (an omega-3 fatty acid) is plant in leafy greens, hemp, flax, soy, walnuts, and canola oil. Other of import omega-3 fatty acids, such equally EPA (eicosapentaenoic acrid) and DHA (docosahexaenoic acid) are found in fatty fish (e.g., salmon, tuna). Linoleic acid (an omega-6 fat acrid) is found in vegetable and nut oils. Because these essential fatty acids are easily accessible, essential fatty acid deficiency is extremely rare.
Phospholipids
Phospholipids make upwards only about ii percent of dietary lipids. They are water-soluble and are found in both plants and animals. Phospholipids are crucial for building the protective barrier, or membrane, around your torso's cells. Although phospholipids are disquisitional, they are not considered to be an essential nutrient because phospholipids are synthesized in the body. In blood and body fluids, phospholipids form structures in which fatty is enclosed and transported throughout the bloodstream.
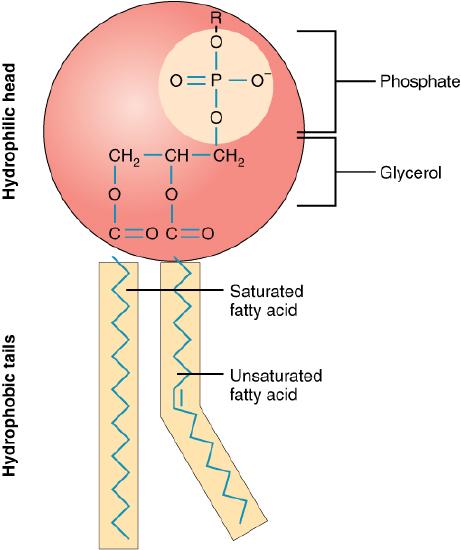
Like triacylglycerols, phospholipids have a glycerol backbone. But unlike triacylglycerols, phospholipids are diglycerides (two fatty-acid molecules attached to the glycerol backbone) while their third fatty-acrid chain has a phosphate group (Figure \(\PageIndex{6}\)). This unique structure makes phospholipids water soluble. Phospholipids are what we call amphiphilic—the fatty-acid sides are hydrophobic (dislike water) and the phosphate group is hydrophilic (likes water).
Phospholipids are platonic emulsifiers that can keep oil and h2o mixed. Emulsions are mixtures of two liquids that practice non mix. Without emulsifiers, the fatty and water content would be somewhat separate within food. Lecithin (found in egg yolk, dear, and mustard) is a popular food emulsifier. When you read most lipid digestion later in this affiliate, yous'll run across the importance of emulsifiers within the body.
In the body, phospholipids bind together to form cell membranes. The amphiphilic nature of phospholipids governs their function as components of cell membranes. The phospholipids form a double layer in cell membranes, thus effectively protecting the inside of the cell from the outside environment while at the same time allowing for transport of fat and water through the membrane.
Sterols
Sterols are the least common type of lipid. Cholesterol is perhaps the nearly well-known sterol and is the most mutual sterol in the nutrition. The trunk produces the majority of its cholesterol; however, a modest corporeality does come up from the diet (from fauna foods). Since cholesterol is produced in our body, it is not essential that we go information technology from our food. Cholesterol is an of import component of the prison cell membrane and is required for the synthesis of sexual activity hormones, vitamin D, and bile salts.
Sterols have a very different construction from triacylglycerols and phospholipids. Most sterols practise not incorporate any fatty acids but rather are multi-ring structures. They are complex molecules that contain interlinking rings of carbon atoms, with side chains of carbon, hydrogen, and oxygen attached. Cholesterol is the best-known sterol because of its function in heart affliction. It forms a large part of the plaque that narrows the arteries in atherosclerosis. Although cholesterol has a bad reputation when it comes to heart wellness, it is a vital substance in the torso that poses a business only when there is excess aggregating of it in the blood.
Central Takeaways
- Lipids include triacylglycerols, phospholipids, and sterols; triacylglycerols are the well-nigh common lipid in food and in the body.
- Three fatty acids combine with one glycerol to brand a triacylglycerol, the body's storage form of fatty.
- The carbon concatenation in fatty acids can be saturated (filled with hydrogen), monounsaturated (with one point of unsaturation and possessing one carbon-carbon double bond), or polyunsaturated (with more than one point of unsaturation and possessing two or more double-carbon bonds).
- The structure and length of a fat acid determines whether it is solid or liquid at room temperature.
- Trans fatty acids have been associated with increased risk for heart illness because of the way they negatively bear on blood cholesterol levels.
- Essential fatty acids cannot be synthesized by the body and must be included in the diet. Omega-3 and omega-half-dozen fatty acids have opposing functions and must exist consumed in proper rest to promote health.
- Phospholipids are diglycerides with a phosphate group in identify of the third fatty-acid chain.
- Sterols comprise a multi-band structure. Cholesterol serves of import body functions such as vitamin D and hormone synthesis and is of import in jail cell membrane structure.
Source: https://med.libretexts.org/Courses/Metropolitan_State_University_of_Denver/Introduction_to_Nutrition_(Diker)/05%3A_Lipids/5.01%3A_What_Are_Lipids
0 Response to "Lipids Are a Family of Compounds That Include"
Post a Comment